Roger Lam
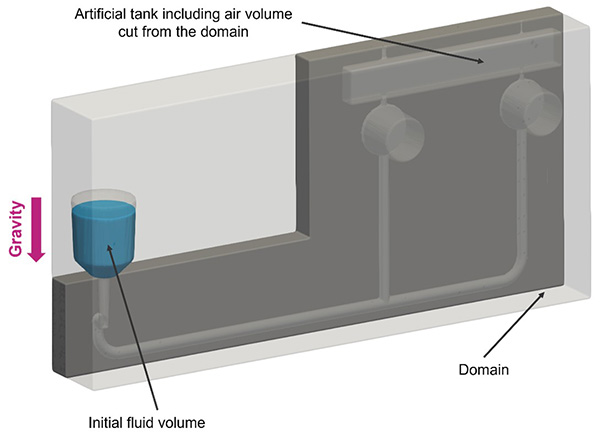
…of POC standard components The Role of Computational Simulations in Holistic POC Design In the realm of computational simulations, POC devices represent a transformative platform for technological advancements. Computational…
…of POC standard components The Role of Computational Simulations in Holistic POC Design In the realm of computational simulations, POC devices represent a transformative platform for technological advancements. Computational…

The FDA is seeking subject matter experts to join its new Digital Health Advisory Committee. The role of the committee will be to support safe and effective regulation of digital…
The FDA is seeking subject matter experts to join its new Digital Health Advisory Committee. The role of the committee will be to support safe and effective regulation of digital…

…to be implemented in the coming years with the proliferation of AI and digital health platforms into our healthcare system, and AdvaMed looks forward to working with lawmakers and the…
…to be implemented in the coming years with the proliferation of AI and digital health platforms into our healthcare system, and AdvaMed looks forward to working with lawmakers and the…

Artificial intelligence (AI) and machine learning (ML) are poised to be play a pivotal role in clinical trials by automating patient recruitment, data collection and analysis. However, most AI algorithms…
Artificial intelligence (AI) and machine learning (ML) are poised to be play a pivotal role in clinical trials by automating patient recruitment, data collection and analysis. However, most AI algorithms…

…Walking is a more natural and automatic activity. I can ask my patients to walk with much less instruction than it takes to complete a different standardized test—which helps differentiate…
…Walking is a more natural and automatic activity. I can ask my patients to walk with much less instruction than it takes to complete a different standardized test—which helps differentiate…

…1061, Rockville, MD 20852. Identify comments with docket number FDA-2012-N-1021 for “Instructions for Submitting Comments on the CDRH Fiscal Year 2024 Proposed Guidance Development.” Submit electronic comments to www.regulations.gov. …
…1061, Rockville, MD 20852. Identify comments with docket number FDA-2012-N-1021 for “Instructions for Submitting Comments on the CDRH Fiscal Year 2024 Proposed Guidance Development.” Submit electronic comments to www.regulations.gov. …