
Despite AI Advancements, Human Oversight Remains Essential
Study reveals limitations of large language AI models in medical coding.

Study reveals limitations of large language AI models in medical coding.

“In the future, CDRH intends to increase the breadth of the dataset of chemicals and RRF information it makes publicly available, and to add other detection methodologies such as liquid chromatography (LC).”

“Companies that operate with business integrity elevate industry standards and perform better in the long-term. We are impressed with Flex’s continued focus on ethical business practices, and dedication to making a real impact for its stakeholders.”

The FDA is hosting it next town hall on medical device sterilization on April 29. The session will include discussion on topics and formats for future sessions in the series.

The consent decree restricts the production and sale of new continuous positive airway pressure (CPAP) machines, bi-level positive airway pressure (BiPAP) machines and other devices at several Philips Respironics facilities in the U.S. until certain requirements are met.

On April 17, the FDA is hosting a Virtual Public Workshop on approaches to Accreditation Scheme for Conformity Assessment Expansion.

“These devices present a number of psychological risks including depression, anxiety, worsening of underlying symptoms, development of post-traumatic stress disorder, and physical risks such as pain, burns, and tissue damage.”

With more than 50 years in the medical device space, SERF SAS is recognized for its innovations in hip implants, including the invention of the original Dual Mobility Cup.
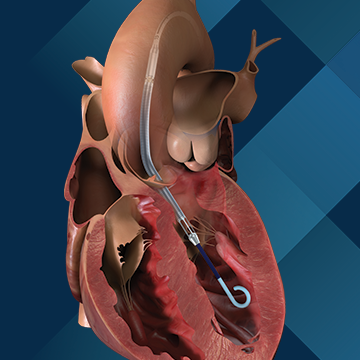
Use of the affected Impella pumps may cause serious adverse health consequences, including left ventricle perforation or free wall rupture, hypertension, lack of blood flow, and death. To date, there have been 129 reported serious injuries, including 49 reports of death.

The Rubitection skin assessment management tool for chronic wounds and Kofimi Technology’s pediatric pulse oximeter device received a combined $50,000 in grant funding as part of the ADPI-led pitch competition.