Across the globe, original equipment manufacturers (OEM’s) are facing tremendous market pressure to create new products in record time. Not only are products expected to maintain the highest quality possible throughout their expected lifecycles, but consumers are also demanding that products do more for less—and they want them now (or as quickly as possible). OEM’s are aggressively pursuing strategies, methodologies, systems and tools that allow their organizations to wield a competitive advantage in the market by offering greater customer value.
To this end, many companies are focused on developing ways to enhance their company’s performance at the very beginning of the new product development process. It is during this “pre-prototype” phase—the earliest and often unmeasured conceptual time period—that the greatest potential for competitive advantage in time to market and cost can be realized. This two-part article will define when pre-prototype begins and ends, explain why it is so important to researchers and organizations alike, and provide some best practices for harnessing the power of pre-prototype to gain competitive advantage.
What is “Pre-prototype”?
“Pre-prototype” is the earliest phase of the new product development process. New product development (NPD) refers to the complete process of bringing a new product to market—whether it is a new, breakthrough technology or a minor, incremental modification to a previously introduced product. The NPD process defines the series of activities, from ideation to market introduction (also called commercialization), through which virtually all products travel. While several conceptual approaches have been proposed to describe the idealized “best” method of developing new products, the most commonly accepted model was developed by Dr. Robert G. Cooper after more than 30 years of studying the practices and pitfalls of more than 30,000 new product introductions. Now implemented by nearly 80% of North American companies, this eight-stage process provides a common framework for structuring and validating significant business decisions at key points throughout the NPD process.1 In addition to keeping members of the development team focused and on the same page, this formal process helps maximize the probability of success and mitigate the risk of failure.
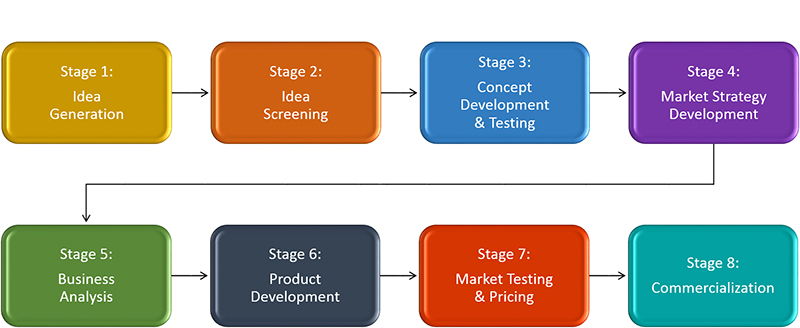
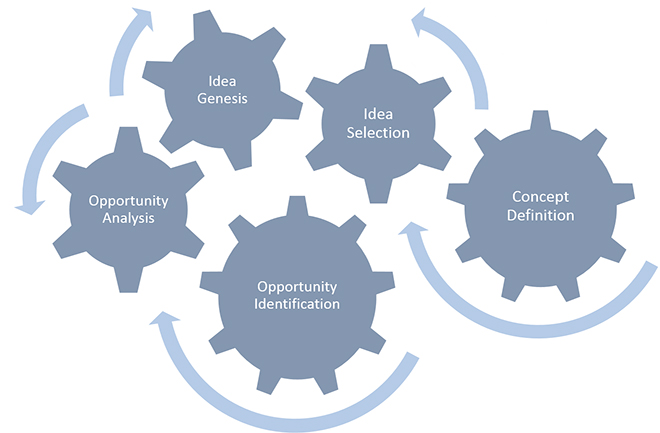
While Cooper’s NPD process may seem familiar to many, less so is the pre-prototype phase, Fuzzy Front End (FFE), or “front end of innovation” of the NPD process. This period consists of the set of activities that occurs at the beginning of the NPD process—before the organization approves and commits to development of a selected concept. In most cases, pre-prototype begins with the spark of an idea, although some may debate that the clock starts even earlier, such as when the market opportunity for a new product arises. In either case, pre-prototype generally encompasses Stage 1(Idea Generation) through Stage 3 (Concept Development & Testing), and it may continue through Stage 6 (Product Development) when a company has typically fully committed to a concept and the design is “frozen.”
Defined by Peter A. Koen as “often chaotic, unpredictable and unstructured”, this preliminary period may take up to 50% of the total product development time and consists of five front-end elements.2
| Opportunity |
A business or technical need that the company or individual realizes by design or default that they might want to pursue to capture competitive advantage, respond to a threat, or solve a problem |
| Idea | Most embryonic form of a new product, service or envisioned solution |
| Concept | Has a defined form (i.e., written and visual) with features and customer benefits combined with a broad understanding of the technology needed |
| Table 1. Pre-Prototype definitions |
Why is Pre-prototype Important?
The power of pre-prototype owes to the fact that at the beginning of the NPD process, the degree of freedom in design and influence on product outcomes are high, whereas the costs for changes are low. This front-end advantage is limited by the fact that the amount and certainty of information is low compared to later stages in the process.
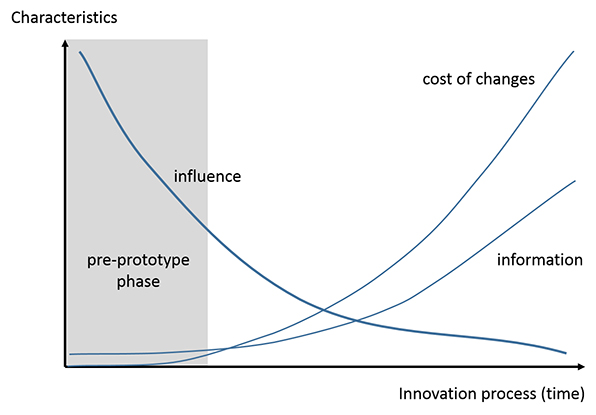
Unfortunately, many companies have insufficient means to formally track or manage these early stages of the NPD process, and the biggest opportunity to save time and money is gone before the clock even starts. It is during this critical upfront time period that a collaborative relationship with a contract manufacturer, a fully capable outsourcing partner, can provide the most value to a company. Part II of this article describes the relationship in detail and the best pre-prototype strategies.
References
- Dr. Scott Edgett and Dr. Robert Cooper Biographies. (1996-2005). Product Development Institute Inc. Retrieved from http://www.prod-dev.com/edgett_cooper.php
- Koen, P.A. (May 25, 2004). Undertstanding the Front End: A Common Language and Structured Picture. Stevens Institute of Technology. Retrieved from http://www.slideshare.net/BrandGenetics/koen-fei