

Companies can improve their chances for a successful transition by leveraging their knowledge of their medical devices and understanding what each device’s categorization under the MDR means to its testing strategy.

Companies can improve their chances for a successful transition by leveraging their knowledge of their medical devices and understanding what each device’s categorization under the MDR means to its testing strategy.

New health-related data is arriving almost daily. Making sense of it all is a huge challenge.

Although medical device manufacturers have more time to prepare due to the delayed EU MDR deadline, this shouldn’t distract from the extensive documentation they must compile in the meantime to prove their devices are compliant.

During product development, collaboration between software, hardware and quality teams must be tracked and seamless to meet the increasing pace of market demand.

In general, the Asian markets have controlled the COVID-19 virus successfully outside of China, but its effect has still led to new developments and trends.

AdvaMed’s Diagnostic Supply Registry is tracking the progress of molecular diagnostic tests to help support state and federal governments.

The authorization is a step toward broad screening that will help reopen schools and workplaces in America, says FDA Commissioner Stephen Hahn, M.D.

With everything to gain, telehealth may lead to a new standard in health/wellness treatment while at the same time unifying the entire healthcare ecosystem.
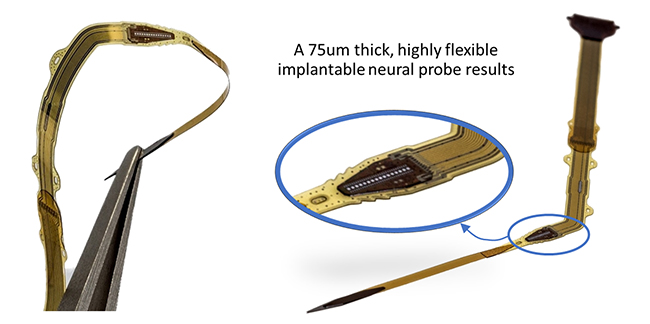
When a device is implanted in a human body, there is always a distinct reaction of the body’s immune system, often resulting in a thick layer of scar tissue surrounding the implant. With device miniaturization and by providing a ‘biomimetic’ device encapsulation, this body reaction will be reduced.

Operating with an antiquated QMS raises your risk. Here’s what to consider when modernizing your system.