Researchers at the National Institute of Standards and Technology (NIST) have developed a tool for building a system known as heart-on-a-chip (HoC). This technology seeks to address the limitations of conventional cardiovascular drug development, which relies heavily on animal testing. By replicating humanlike models to study cardiovascular diseases, NIST believes the HoC system has the potential to replace animal testing, shorten drug development timelines and reduce costs.
The HoC device mimics the intricate interactions of cells within the heart on a small chip and is part of the larger organ-on-a-chip (OoC) suite. The actual design of the HoC varies, but it is typically a small, transparent or semi-transparent chip consisting of a network of microchannels printed on a layer of polymer. These microchannels are designed to imitate the blood vessels found in the human heart. Researchers place human heart cells within these microchannels to manipulate and observe their behavior. Researchers can independently stimulate them or observe their behavior under different conditions, such as the introduction of a drug.
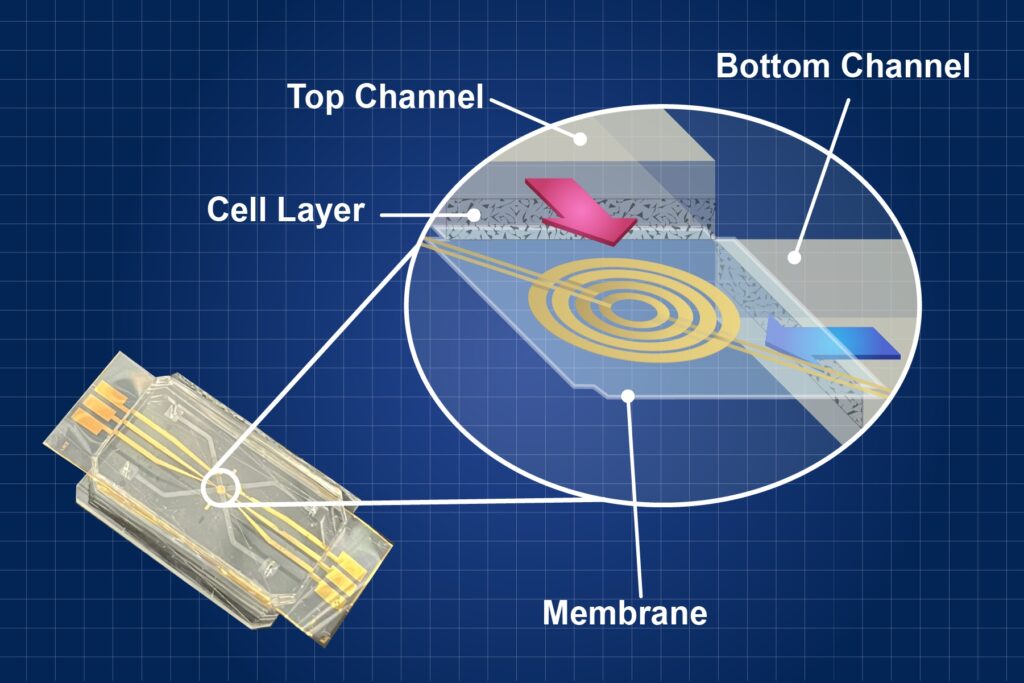
“The heart-on-a-chip is designed to mimic the conditions of a real heart,” said NIST researcher Darwin Reyes, who led the development of this HoC system. “We can manipulate the environment to change stem cells into heart cells and make them contract and relax, as they do in a body to produce a heartbeat.”
The “heart” of the organ-on-a-chip system lies in something called microfluidics, which is essentially a miniature plumbing system in which researchers can precisely control and manipulate tiny amounts of liquids. Researchers use microfluidics to create advanced models of organs and tissues on small chips in the lab.
“The chip itself can be used with many different cell types,” said Reyes. “In this project in particular, we’re using heart cells, but you can use a customized version of the system with other cells to monitor their behavior visually and electronically.”
Researchers can create chips that mimic the conditions of various organs, and these chips can even be interconnected to form a multi-organ system.
A Superior Alternative to Animal Testing
Researchers at NIST see a significant benefit in using the chips to replace animal testing, as animal physiology does not perfectly match human physiology, which can both delay the drug testing process and put human test subjects at risk of adverse effects from the drug. Additionally, there is ongoing debate about the ethical considerations of animal testing.
“The ultimate goal is to, if possible, be able to skip the animal testing altogether,” said Reyes. “This would also shorten the time it takes to test drugs, which would hopefully make the medications cost less.”
In 2022, President Joe Biden signed into law the FDA Modernization Act 2.0. The bill essentially revises the Federal Food, Drug, and Cosmetics Act of 1938, which mandated animal testing for every new drug development protocol. While for the past century, the mandate was intended to ensure certain quality and safety standards for drugs and medical devices, recent advancements in science have begun to offer increasingly viable alternatives to animal testing, including organ-on-a-chip systems.







