
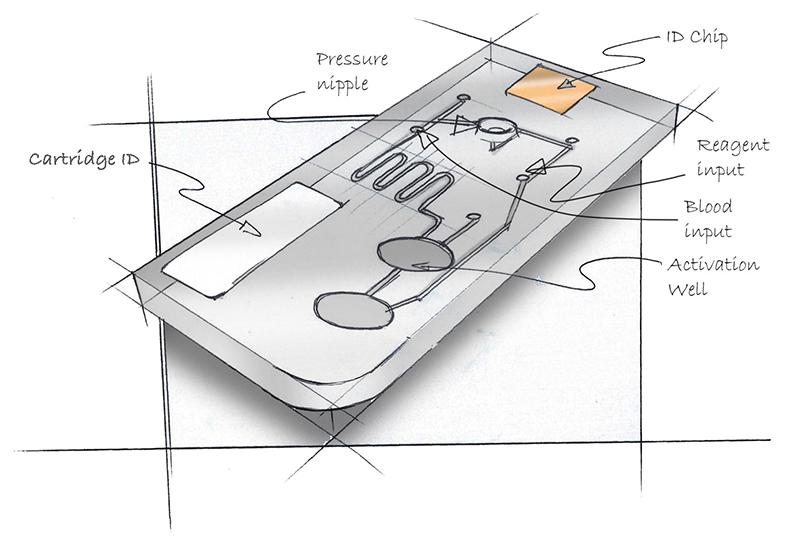
By examining the larger user experience involved in the microfluidics process, we can identify issues, and design these experiences to reduce the potential for user error, improve outcomes and create a simpler, more accessible process.

By examining the larger user experience involved in the microfluidics process, we can identify issues, and design these experiences to reduce the potential for user error, improve outcomes and create a simpler, more accessible process.

Modern technology has given rise to new legal questions. How does FDA regulate machine-learning computers that are changing so rapidly – given that the approved product may be drastically different than the product that ends up on the market? These questions arise from a lack of understanding of the complex nature of AI/ML-based SaMD, the opaqueness of the regulatory framework, and a dearth of relevant case law.

In this new survey, medtech manufacturers are invited to share their perspective on business initiatives, manufacturing execution systems and latest challenges.

It is critical for leaders in the medtech space to ensure that nonviable medical devices and equipment are destroyed properly, while focusing on long-term sustainability goals.

Medical devices and medical software are becoming increasingly connected to hospital networks, other medical devices or the Internet. As a result, manufacturers and developers are required to consider cybersecurity from the very early stages of development. This in turn necessitates comprehensive risk management along the entire lifecycle of a device.
The increasing incidence of hearing loss and neurological diseases has prompted the development of convenient and enhanced solutions in wearable healthcare devices.
Diagnostic advances bode well for the imminent expansion of the global COVID-19 detection kits market, which is expected to be worth $8 billion by 2027.

Greater use of next generation sequencing can better prepare the world for future diseases and accelerate the adoption of precision care.
Adhesive selection can make or break the success of a stick-to-skin wearable device. This article reviews six questions to ask the next time you design a device intended for wear on fragile skin.

To change the landscape of cyber threats in healthcare, companies must prioritize designing security into devices.